On needs to know about the condition of heart failure. It is a condition in which the patient’s heart is not in a condition to pump the blood to meet the needs of the body. There are certain cases in which the heart of the person is unable to fill the enough blood and in other cases the heart is not able to pump the blood to other parts of body with the force that is required. It can’t be said when there is heart failure, the heart has stopped working. But the heart failure is a condition that requires much medical care. It develops with a passage of time as the pumping action of heart gets weaker. Either the right side of the heart can be affected or both the sides but as seen in most of the cases it involves in both sides of the heart.
There is certain indication which tells about the heart failure that are:
When the risk of the mortality is greater than 80% then ECLS should be considered.
PaO2/FiO2<150 on FiO2>90% is one of the things that is associated with the 80% mortality risk.
Syndromes like severe air leak.
The person’s cardiac or respiratory collapsed immediately.
If a person is in a list of lung transplant that needs intubation.
- One thinks about the contradiction with the heart failure but there are no contradictions that are related to ECLS, as each and every patient is consider as a separate individual with the respect of risks and benefits. Some of the conditions are there which are associated with the poor outcome and it can also be considered as the relative contradictions that includes:
After successful LVAD implantation, you need to adjust to a new kind of life. You need to be connected to a power source and external controller at all the times. The devise runs on electrical power and will be connected to charging while you are sleeping. An extra controller along with a fully charged battery is kept in case of an emergency. You need to carry the backup whenever you leave home. Also you will have to stay away from kids and pets. It's a great aspect of latest devises that you will be able to move freely and enjoy moderate intimacy.
More than 28,000 devices have been implanted worldwide and currently 10,000+ patients are on the device at present.

Dr Kewal Krishan is amongst few esteemed cardio theraic surgen who holds expertise in all aspects of heart transpiration including LVAD () He is the one with most number of LVAD in Delhi
With the help of the advanced technology, we use high tech therapies in order to treat the patients who suffer from the heart disease and end stage heart failure
Contact for any query: Heart Failure Coordinator:- 9540-500-800 (helpline)
Mechanical support:
Indications:
Post cardiotomy failure
Acute myocardial infarctionwhohave undergone such significant myocardial damage that recoveryis unlikely.
Acute decompensation of chronic failure
Acute myocarditis,
Chronic heart failure in transplantation candidates
Intractable ventricular arrhythmias not responding to cardiac defibrillators and aggressive pharmacologicregimens
High-risk reparative cardiac operation
Terminology and Classification of MCS Devices
- Ventricle Supported
- Left ventricular assist device
- Right ventricular assist device
- Biventricular assist device
- Total artificial heart
- Intended Use
- Short-term MCS
- Patient remains within hospital eg. Intraaortic balloon pump, Tandem heart, Centrimag, Impella ,Abiomed BVS 5000
- Long-term, “Durable” MCS
- Patient discharged to home – “hands free” untethered mobility eg.ThoratecHeartMate I1 MicroMedDeBakeyHeartAssist 5Jarvik 2000Berlin HeartIncor, HeartWareHVAD,DuraHeart
- Bridge to recovery
- Bridge to transplant
- Destination therapy
Intraoartic balloon pump,TandemHeartpVAD, AbiomedImpella 2.5 LP provide partial circulatory support (15-30%). Other short and long term devices provide full circulatory support.
TandemHeartpVAD (Percutaneous left atrial-to-femoral-arterial VAD)
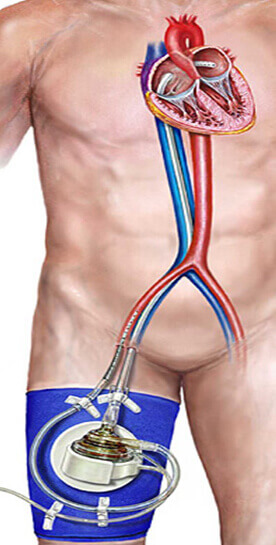

Figure 3. Tandem Heart pVAD
It is Continuous-flow centrifugal pump which provides partial sup
port, average flow is usually 3.5 to 4.0 L/min at 7500 RPM. The cannulae are placed percutaneously under fluoroscopy. The 21 french (F) left atrial drainage cannula through transeptal approach and 15 to 17 F femoral artery cannula are used. The role of this device for short-term stabilization as bridge to recovery or as a bridge to definite surgical treatment
IMPELLA :

Figure 4. Surgical placement of Impella device.
Figure 5.Percutaneous placement of Impella device.
Impellamicroaxial flow device, which is a miniature impeller pump located within a catheter. Impella was designed for either surgical placement via a graft in the ascending aorta (5.0 LD) or for percutaneous placement (2.5 L )via the femoral artery (13,14).It provides partial to full support.(Figure 4 & 5)
CENTRIFUGAL PUMPS :

Figure 8.The Thoratec ventricular assist
system in the biventricular support
configuration

Figure 9. LevitronixCentrimag®
centrifugal pump
Centrifugal pumps are an extension of cardiopulmonary bypass. They use rotating cones or impellers to generate energy that is recovered in the form of pressure flow work. There are presently three centrifugal pumps available, the Bio- Medicus (Bio-MedicusInc, Minneapolis, MN), the Sarns (Sarns/3M Ann Arbor, MI) and the LevitronixCentrimag® (Levitronix LLC, Waltham, MA) (Figure 5). All of them have the capability of supporting patients who cannot be weaned from cardiopulmonary bypass or who are waiting cardiac transplantation. The pumps are versatile and can be used as a right ventricular assist device (RVAD), left ventricular assist device (LVAD) or biventricular (BiVAD) support.
CentriMag or other short term devices are frequently used mainly for the following reasons.
- Left ventricular failure post cardiotomy or transplant graft failure
- Right ventricular failure post cardiotomy
- Right ventricular failure post LVAD insertion
- Myocarditis
- Post infarction shock
- As ECMO support
- Bridge to a decision, whether it be to attempt recovery, long-term LVAD, or transplant
EXTRACORPOREAL MEMBRANE OXYGENATION (ECMO) :
This is mainly used when pulmonary support is required with or without systemic support. It removes carbon dioxide from and adds oxygen to venous blood via an artificial membrane lung. The pulmonary circulation is bypassed, and oxygenated blood returns to the patient via an arterial or venous route. With veno-venous bypass, ECMO is effective primarily as a therapeutic option for patients with severe respiratory failure. With veno-arterial bypass, an extracorporeal pump is employed to support systemic perfusion, thus providing a hemodynamic support option in patients with cardiac failure. ECMO is a temporary life support for patients with potentially reversible severe acute respiratory failure or cardiac failure (15).
1. Pulsatile Volume Displacement pump (First Generation Pumps) :

Figure 11.Heart Mate XVE
Out of first generation pumps Heart Mate XVE is the most commonly used device.It was the first device used for REMATCH trial which showed benefit of device placement over medical therapy.This device is also approved by FDA for the destination therapy.Now a days this is being used less frequently because of cumbersome size, limited durability and availability of smaller and more durable pumps.
2. Continuous-flow Rotary Pumps (2nd and 3rd Generation pumps):

Figure 12 .HeartMate II
The second generation pump most commonly used is Heart Mate II axial flow pump.The third generation pumps are magnetically levitated centrifugal blood flow pumps with no contact bearing design.
Continuous-flow Rotary Pumps
Advantages:
- No heart Valves
- No flexible diaphragm
- No large housing to accommodate pump "stroke volume"
- Fewer moving parts
- Potential for biventricular application
Disadvantages:
- Minimal pulse
- Significant occurs
- Operated in fixed mode speed may limit cardiac output
- After load dependent
Heart Mate II : is a 2nd generation axial flow pump Most commonly implanted device.Its durability, reliability and promising results opened the avenue for mechanical support. Its potential for small size, low noise, and absence of a compliance chamber, have been developed for clinical use. It provides continuous rather than pulsatile flow and are totally implantable.
Magnetically levitated centrifugal pumps : These are currently undergoing clinical trials for the treatment of heart failure. They have several advantages over the axial flow pumps: 1) they are energetically more efficient 2) they have lower tolerances so manufacturing is easier and they are less prone to thrombosis 3) they are potential very durable (>10 year life-span). The three main devices in this category are the VentracorVentrAssist LVAD, the Heartware LVAD and RVAD and the Terumo Duraheart.
HeartWareHVAD :

Figure 13 HeartWare
pump
This is a third generation centrifugal pump.
It weighs 145 grams and pumps 4 to 5 liters of blood per minute.This does not requires pocket formation unlike other devices
CIRCULITE :


Figue 14 Circilite device and its placement.
CircuLite’s circulatory support systems are designed with unique attributes, which include:
World's smallest implantable blood pump (the size of a AA battery and weighing 25g) First superficially implanted micro-pump, designed to be placed without cardiopulmonary bypass or sternotomy Small percutaneous lead Designed to supplement native cardiac output Designed for long-term support Magnetically and hydrodynamically stabilized rotor designLightweight, rechargeable dual battery pack system (average power 6-8 hours)
With the immutable limitation in the supply of suitable donor hearts a lot of patients with heart failure could not be offered the possibility of long survival and in the last 10- 15 years were developed a second and third generation of pumps. Those patients who do not qualify for transplantation long term devices are used for them as a destination therapy. In addition, destination recipients can undergo heart transplantation after their relative contraindications improves on mechanical support. The vast majority of deaths occur within the first 3 months after LVAD surgery. Sepsis, right heart failure and multi organ failure are the main causes of postoperative death and are the main contributors to the relatively high in-hospital mortality after device implantation.
TOTAL ARTIFICIAL HEART

Figure 15. Total artificial heart
SynCardia total artificial heart
The SynCardia total artificial heart (TAH) is a pulsatile, biventricular, pneumatically driven, orthotopic TAH that replaces some of the recipient atrial tissue, all 4 cardiac valves, both ventricles, and the proximal portions of the aorta and pulmonary arteries (16).
Each ventricle has a polyurethane diaphragm that separates blood from air. Medtronic-Hall mechanical valves in the atrioventricular and the semilunar positions (27 mm inflow and 25 mm outflow) provide unidirectional blood flow. Maximum stroke volume is 70 mL, and maximum cardiac output is 9.5 L/min. Total weight is 160 g with a volume displacement of 400 mL. A pneumatic driveline is connected to each ventricle, which is tunneled through the chest wall and connected to an external console.
AbioCor total artificial heart
The AbioCor TAH is a fully implantable, biventricular, orthotopic, pulsatile electrohydraulic TAH that is driven by a battery-powered motor. The motor powers an internal centrifugal pump, which hydraulically moves a membrane sac responsible for the pumping action.TheAbioCor TAH replaces recipient atrial tissue, all 4 cardiac valves, both ventricles, and the proximal portions of the aorta and artery. The pump and valves are made from titanium and proprietary polyurethane. A miniaturized internal electronics package monitors the pumping speed (17).
An internally implantable battery is continuously recharged from an external power source by a process called transcutaneous energy transmission (TET), which obviates the need for percutaneous power or pneumatic drive lines. An external battery pack with a TET coil provides 2-4 hours of use, while the internal battery allows 30 minutes of tether-free operation.TheAbioCor TAH weighs 1090 g and has a volume displacement of 800 mL, making it significantly larger and heavier than the SynCardia TAH.
Now a days TAH is being used more frequently and patients can be sent home with portable driver.The main advantage of TAH is it provides biventricular support in orthotopic position.











